dPCR Market to Surpass US$14.34 Billion by 2031, Propelled by Escalating Demand for High‑Precision Genetic Analysis – New Industry Research by The Insight Partners.

Growing demand for sensitive and accurate nucleic acid detection, wider use in clinical diagnostics and personalized medicine, and ongoing advances in dPCR technology are driving demand for dPCR.
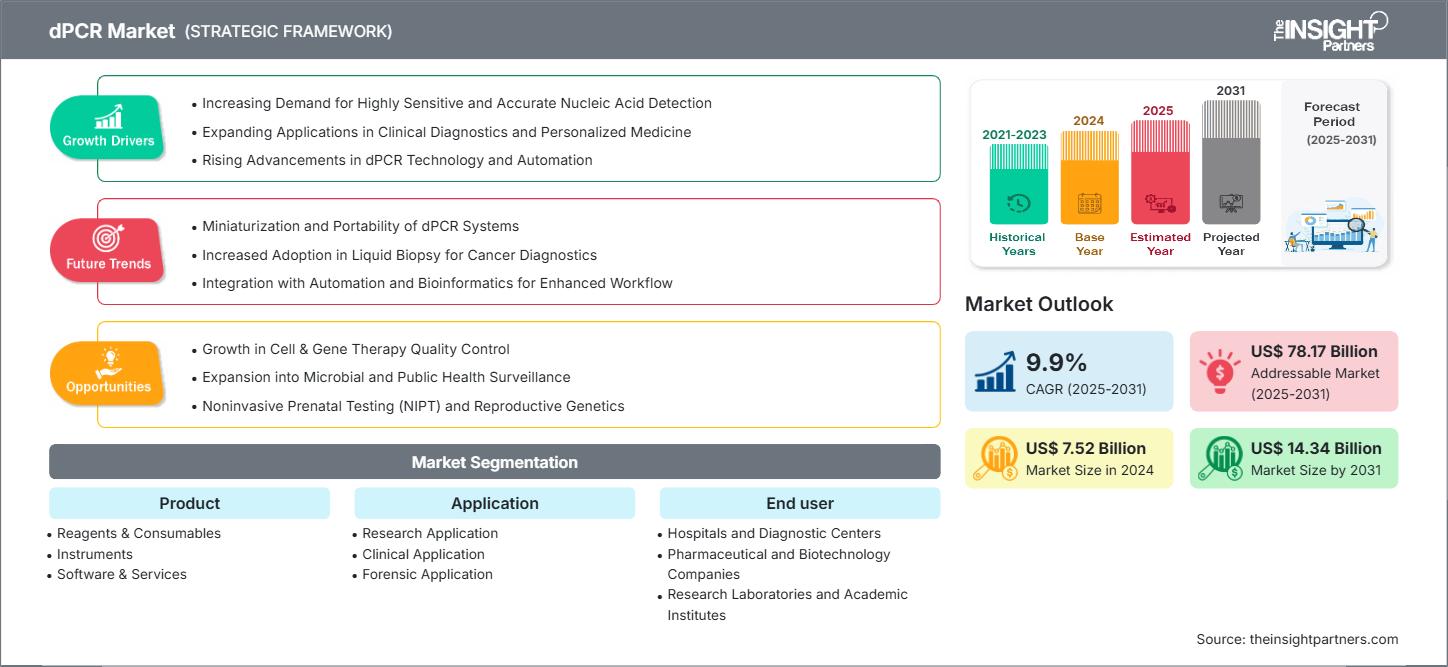
New York, NY, April 30, 2026 (GLOBE NEWSWIRE) — A new comprehensive market research report from The Insight Partners reveals that the Global dPCR Market is projected to grow from US$ 7.52 billion in 2024 to US$ 14.34 billion by 2031, registering a CAGR of 9.4% during the forecast period from 2025 to 2031.
Market Growth Highlights:
- The Digital PCR (dPCR) market in North America is expanding at a CAGR of 10.3% and is one of the fastest-growing segments within molecular diagnostics, outpacing major traditional PCR technologies. Its ability to detect low-abundance mutations and its expanding applications in infectious disease testing are projected to boost the growth of dPCR market during the forecast period.
- The dPCR market in the United States is anticipated to expand at a CAGR of 10.7% during 2025-2031. dPCR is transitioning from a capital research tool to mainstream clinical diagnostic platform in the US. The market is expanding because of rising needs for accurate genetic measurement, increasing application in clinical diagnostics and personalized medicine, and infectious disease monitoring, and the substantial funding dedicated to life sciences research
- Primary Growth Driver: Key drivers of the dPCR market include rising demand for precise genetic analysis, growing clinical diagnostics, infectious disease detection, and advancements in molecular biology technologies.
- Key Segment:Clinical Application remains dominant, while Research Application exhibits the highest CAGR (11.3%).
Download Sample PDF @https://www.theinsightpartners.com/sample/TIPRE00042218/
Clinical and research facilities are adopting advanced molecular diagnostics, which provide high detection accuracy. The CDC Behavioral Risk Factor Surveillance System reported that in 2023, approximately 76.4% of American adults, which represents 194 million people, had at least one chronic condition.
The sustained growth of the dPCR market reflects incremental adoption as well as a shift toward ultra-sensitive and quantitative molecular testing. Clinical labs and research centers demand digital PCR for accurate nucleic acid measurement because of the increased prevalence of genetic disorders, infectious diseases, and cancer. dPCR enables people to detect diseases at an early stage, track minimal residual disease, and implement precision oncology methods. Academic institutions and biotech companies use dPCR to perform advanced genetic testing. Bio-Rad Laboratories and Thermo Fisher Scientific provide dPCR systems to fulfill clinical and research requirements.
Market Momentum Driven by Expanding Applications In Clinical Diagnostics and Personalized Medicine.
Rising Advancements in dPCR Technology and Automation:
The increasing use of dPCR technology for cancer detection, infectious disease testing, prenatal assessments, and gene therapy applications is driving demand for precise measurements of nucleic acids. Laboratories achieve better operational results by using automated systems that combine robotics with high-throughput testing equipment. Experimental results improve through cloud data storage, artificial intelligence analytics, and immediate data observation. Companies are developing multiplexed systems, microfluidic cartridges, and biocompatible reagents to expand applications while maintaining sample integrity and assay precision.
New possibilities arise from personalized medicine, biomarker discovery, and low-level pathogen detection. The developing areas adopt molecular diagnostics through increased funding for research and the establishment of research partnerships between biotech companies and academic institutions. The industry faces three main challenges: high equipment costs, regulatory requirements, and the complexity of advanced technologies. The market maintains its momentum through ongoing technical improvements, better test tools, and next-generation sequencing technology. Competitive dynamics drive companies to develop new products, establish partnerships, and create distribution systems across countries.
Request a customized report to align these insights with your strategic business objectives
Major dPCR Companies Include:
- Abbott
- Qiagen
- Bio-Rad Laboratories Inc.
- Agilent Technologies, Inc.
- Thermo Fisher Scientific, Inc.
- GE Healthcare
- bioMérieux
- Hoffmann-La Roche Ltd
- Takara Bio Inc
- Stilla Technologies.
Explore E-insights – The Expert market intelligence platform developed by The Insight Partners. A Platform that offers impactful Market Intelligence at your fingertips to support your revenue goals and have a competitive edge. Helps you accomplish objectives
dPCR Market Segmentation Analysis:
Clinical Application Segment to Grow at the Highest CAGR
dPCR technology provides precise measurement of small amounts of genetic material, which enables early cancer mutation detection, minimal residual disease monitoring, and high-sensitivity viral pathogen identification. The demand for personalized treatment methods and biomarker-based therapies drives hospital and diagnostic laboratory requirements. The combination of automated workflows with artificial intelligence-based clinical analysis tools improves reproducibility and operational efficiency, which leads to increased use of dPCR technology in healthcare facilities.
Reagents & Consumables to Hold the Largest Market Share
Laboratories require high-quality PCR kits together with enzymes, primers, probes, and microfluidic consumables to conduct precise genetic tests. Clinical laboratories, research institutions, and pharmaceutical companies use these consumables for their work in oncology, infectious disease detection, prenatal testing, and gene therapy. The development of pre-formulated kits together with high-fidelity enzymes and multiplexing capabilities has improved the sensitivity and reproducibility of assays while streamlining laboratory operations. The continuous need for consumables, together with the increasing volume of molecular diagnostic tests and research work, establishes this segment as the market leader.
Asia Pacific to Witness the Fastest Growth:
APAC is witnessing the fastest growth in the digital PCR (dPCR) market, supported by:
• Expanding adoption of molecular diagnostics and genetic testing in China, India, and Japan
• Rising prevalence of cancer, infectious diseases, and genetic disorders
• Increasing awareness of precision medicine and early disease detection
China’s public health programs support genomic research and clinical diagnostic systems. India uses its government healthcare programs to support hospitals and research centers with molecular testing. Countries in Southeast Asia are building laboratory facilities to meet the rising demand for oncology and infectious disease testing. Urban and semi-urban laboratories are increasing their use of dPCR platforms, which enable them to execute advanced testing procedures.
dPCR Market Dynamics:
Driver: Demand For Highly Sensitive and Accurate Nucleic Acid Detection
There is an increasing need for precise and sensitive nucleic acid detection solutions that can be used in clinical diagnostics, research activities, and pharmaceutical research. Digital PCR enables researchers to measure DNA and RNA quantities with complete accuracy, enabling the detection of early cancer mutations, the tracking of residual disease, and the identification of extremely rare pathogens with high accuracy. The growth of personalized medicine, infectious disease testing, prenatal screening, and gene therapy applications continues to drive market expansion. The laboratory and research institution adoption of dPCR solutions increases because automated workflows, multiplexing, and microfluidic platforms help improve assay performance, test results, and operational efficiency.
Opportunity: Growth in Cell & Gene Therapy Quality Control
Digital PCR enables researchers to obtain exact measurements of nucleic acids which allows them to determine the amounts of viral vectors and transgene expression together with residual host cell DNA present in gene therapy products. Manufacturers turn to dPCR testing because it helps them maintain consistent production quality while testing product strength and detecting any potential contamination. The therapeutic sector is expanding because companies invest in advanced biologics and quality standards demand dPCR-based solutions.
Challenge: Lack of Standardization and Regulatory Guidance
Regulatory bodies have not yet developed standardized frameworks for validating and approving dPCR-based diagnostics, especially in the context of clinical and therapeutic applications. Therefore, it is essential to resolve the challenges related to standardization and regulatory guidance to ensure the reproducibility of the results obtained in the process.
Click to Purchase the full report from The Insight Partners
Recent Developments:
- In July 2025, Bio-Rad Laboratories, Inc., a global leader in life science research and clinical diagnostics products, announced the launch of four new Droplet Digital PCR (ddPCR) platforms. The newly introduced instruments include Bio-Rad’s QX Continuum ddPCR system along with the QX700 series of ddPCR platforms acquired as part of the company’s recently completed acquisition of digital PCR developer Stilla Technologies.
- In June 2025, QIAGEN and GENCURIX, Inc. announced a new partnership to develop oncology assays for use on the QIAcuityDx platform, a high-performance digital PCR system designed for clinical diagnostics.
Related Reports:
The Home Diagnostics Market size is expected to reach US$ 12.01 Billion by 2034 from US$ 7.52 Billion in 2025. The market is estimated to record a CAGR of 5.35% from 2026 to 2034.
The Cardiac Diagnostics Catheters Market size is expected to reach US$ 7.38 Billion by 2034 from US$ 3.81 Billion in 2025. The market is estimated to record a CAGR of 7.63% from 2026 to 2034.
The Veterinary Point-of-Care Diagnostics Market size is expected to reach US$ 6.94 Billion by 2034 from US$ 2.89 Billion in 2025. The market is estimated to record a CAGR of 10.21% from 2026 to 2034.
The PCR and NGS-Based Molecular Diagnostics Market size is expected to reach US$ 14.36 Billion by 2034 from US$ 5.85 Billion in 2025. The market is estimated to record a CAGR of 10.49% from 2026 to 2034.
The Neurodiagnostics Market size is expected to reach US$ 26.18 Billion by 2034 from US$ 11.9 Billion in 2025. The market is estimated to record a CAGR of 9.15% from 2026 to 2034.
The global Cardiovascular Disease Diagnostics Market size is projected to reach US$ 26.65 billion by 2034from US$ 12.89 billion in 2025. The market is anticipated to register a CAGR of 9.50% during the forecast period 2026-2034.
The global Oncology Based Molecular Diagnostics Market size is projected to reach US$ 3.28 billion by 2034from US$ 2.4 billion in 2025. The market is anticipated to register a CAGR of 4.02% during the forecast period 2026-2034.
About The Insight Partners
The Insight Partners is a global market research and consulting firm that delivers in-depth analysis and actionable insights across technology, media, telecommunications, healthcare, and industrial sectors. The firm supports organizations in identifying growth opportunities, understanding market dynamics, and making informed strategic decisions.
Contact Us:
If you have any queries about this report or if you would like further information, please contact us:
Contact Person: Ankit Mathur
E-mail: [email protected]
Phone: +1-646-491-9876
Press Release –https://www.theinsightpartners.com/pr/dpcr-market
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. NewIndiaObserver.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. NewIndiaObserver.com takes no editorial responsibility for the same.