Sepsis Clinical Trial Pipeline Appears Robust With 25+ Key Pharma Companies Actively Working in the Therapeutics Segment | DelveInsight

The sepsis clinical trial analysis report delivers important insights into ongoing research of 30+ pipeline sepsis drugs, clinical strategies, upcoming therapeutics, and commercial analysis.
New York, USA, May 12, 2026 (GLOBE NEWSWIRE) — Sepsis Clinical Trial Pipeline Appears Robust With 25+ Key Pharma Companies Actively Working in the Therapeutics Segment | DelveInsight
The sepsis clinical trial analysis report delivers important insights into ongoing research of 30+ pipeline sepsis drugs, clinical strategies, upcoming therapeutics, and commercial analysis.
DelveInsight’s ‘Sepsis Pipeline Insight 2026’ report provides comprehensive global coverage of pipeline therapies for sepsis across various stages of clinical development. The report offers an in-depth analysis of key trends, emerging therapies, and competitive landscape dynamics, highlighting the strategies of major pharmaceutical companies to advance the pipeline and capitalize on future growth opportunities. In addition, it includes critical insights into clinical trial benchmarking, partnering and licensing activities, and regulatory pathways involving the FDA and EMA, enabling stakeholders to make informed decisions and optimize development strategies within the sepsis domain.
Sepsis Clinical Trial Analysis Summary
- DelveInsight’s sepsis pipeline report depicts a robust space with 25+ active players working to develop 30+ pipeline sepsis drugs.
- Key sepsis companies, such as Vivacelle Bio, Inotrem, Shanghai Yidian Pharmaceutical Technology Development, Sobi, AUROBAC Therapeutics, Aptahem AB, Novartis Pharmaceuticals, Guard Therapeutics, Bayer, Artcline GmbH, AstraZeneca, Op-T LLC, Comanche Biopharma, and others, are evaluating new sepsis drugs to improve the treatment landscape.
- Promising pipeline sepsis therapies, such as VBI-S, Nangibotide, YD0743, Emapalumab, ATX101, Apta-1, TIN816, RMC-035, BAY3389934, ARTICE, AZD4144, OPT101, CBP-4888, and others, are in different phases of sepsis clinical trials.
- Approximately 7+ sepsis drugs are in the late stages of development.
- Notable MoAs in sepsis clinical trials include Nitric Oxide Redistribution, TREM-1 inhibitor, Interferon gamma inhibitor, Immunomodulators, CD39 Antagonist, and others.
Request a sample and discover the recent advances in sepsis drug development @ https://www.delveinsight.com/sample-request/sepsis-pipeline-insight
What is Sepsis?
Sepsis is a life-threatening medical condition that occurs when the body’s response to an infection becomes dysregulated, triggering widespread inflammation and organ dysfunction. It typically begins with a bacterial, viral, or fungal infection, commonly in the lungs, urinary tract, abdomen, or bloodstream, but instead of remaining localized, the immune system releases chemicals that can damage tissues and organs throughout the body. As sepsis progresses, it can lead to reduced blood flow, clotting abnormalities, and ultimately organ failure, a severe stage known as septic shock. Early symptoms may include fever, rapid heart rate, confusion, and difficulty breathing, and prompt diagnosis and treatment with antibiotics, fluids, and supportive care are critical to improving survival outcomes.
Find out more about sepsis drug development @ Sepsis Treatment
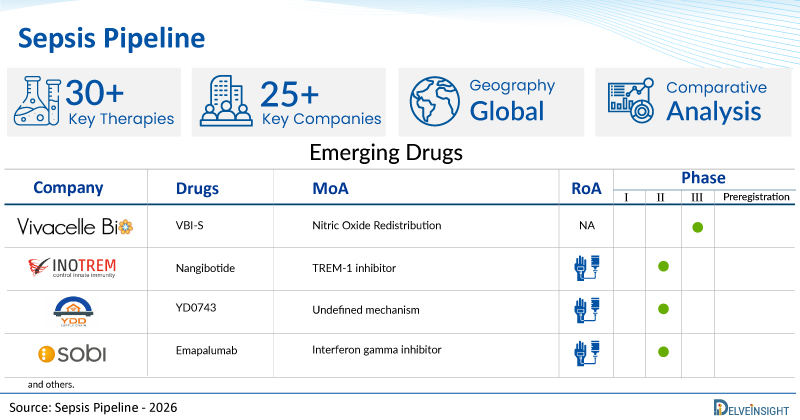
A snapshot of the Pipeline Sepsis Drugs mentioned in the report:
| Drugs | Company | Phase | MoA | RoA |
| VBI-S | Vivacelle Bio | III | Nitric Oxide Redistribution | NA |
| Nangibotide | Inotrem | II | TREM-1 inhibitor | Intravenous |
| YD0743 | Shanghai Yidian Pharmaceutical Technology Development | II | Undefined mechanism | Intravenous Infusion via micro-infusion pump |
| Emapalumab | Swedish Orphan Biovitrum AB (Sobi) | II | Interferon gamma inhibitor | Intravenous |
| ATX101 | AUROBAC Therapeutics | I | Undefined mechanism | Intravenous |
| Apta-1 | Aptahem AB | I | Inhibit protein–protein interactions | Intravenous |
Learn more about the emerging sepsis therapies @ Sepsis Clinical Trials
As per Stuti Mahajan, consulting manager at DelveInsight, sepsis remains a high unmet-need indication with significant mortality, where pipeline focus is shifting toward immunomodulatory and precision medicine approaches, but clinical translation remains challenging due to heterogeneity in patient response and lack of robust biomarkers. The rising prevalence of Sepsis worldwide is a significant driver for the market. The disease is reaching epidemic proportions due to sedentary lifestyles, unhealthy diets, and an aging population. As the number of people with Sepsis grows, the demand for effective treatments and management options increases.
Recent Developments in Sepsis Treatment Space
- In March 2026, AUROBAC announced the successful completion of its Phase I clinical trial (NCT07107802) evaluating ATX101. ATX101 is a potential first-in-class therapy designed to address shock, organ failure, and mortality associated with sepsis. Completion of the Phase I trial marks an important milestone for AUROBAC and reflects the strength of its strategic partnership with Boehringer Ingelheim.
- In January 2026, A new drug developed by Australian researchers showed promising results in reducing sepsis in a Phase II clinical trial involving 180 patients. The carbohydrate-based treatment works by calming a dangerous immune reaction that can cause organ failure. With no specific anti-sepsis therapy currently available, the findings mark a major step forward. Researchers now aim to move into Phase III trials.
- In January 2025, Partner Therapeutics, Inc. announced that they would be funding a phase II study in sepsis patients to evaluate the safety and allow dose selection of LEUKINE (sargramostim, rhu GM-CSF) in partnership with the Biomedical Advanced Research and Development Authority (BARDA), which is a division of the Administration for Strategic Preparedness and Response (ASPR) at the U.S. Department of Health and Human Services.
- In April 2024, AdrenoMed AG announced that their primary product candidate, Enibarcimab, a first-in-class non-neutralizing monoclonal antibody, had been given Fast Track designation by the US Food and Drug Administration (FDA) for the treatment of septic shock.
- In January 2024, an agreement for a Phase III Registration Trial of Nangibotide in Septic Shock had been successfully reached between Inotrem and the FDA.
Scope of the Sepsis Pipeline Report
- Coverage: Global
- Sepsis Therapeutic Assessment By Product Type: Mono, Combination, Mono/Combination
- Sepsis Therapeutic Assessment By Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
- Sepsis Therapeutics Assessment By Route of Administration: Oral, Intravenous, Subcutaneous, Parenteral, Topical
- Sepsis Therapeutics Assessment By Molecule Type: Recombinant fusion proteins, Small molecule, Monoclonal antibody, Peptide, Polymer, Gene therapy
- Sepsis Therapeutics Assessment By Mechanism of Action: Nitric Oxide Redistribution, TREM-1 inhibitor, Interferon gamma inhibitor, Immunomodulators, and others
- Key Sepsis Companies: Vivacelle Bio, Inotrem, Shanghai Yidian Pharmaceutical Technology Development, Sobi, AUROBAC Therapeutics, Aptahem AB, Novartis Pharmaceuticals, Guard Therapeutics, Bayer, Artcline GmbH, AstraZeneca, Op-T LLC, Comanche Biopharma, and others.
- Key Sepsis Pipeline Therapies: VBI-S, Nangibotide, YD0743, Emapalumab, ATX101, Apta-1,TIN816, RMC-035, BAY3389934, ARTICE, AZD4144, OPT101, CBP-4888, and others.
Dive deep into rich insights for new sepsis treatments, visit @ Sepsis Drugs
Table of Contents
| 1. | Sepsis Pipeline Report Introduction |
| 2. | Sepsis Pipeline Report Executive Summary |
| 3. | Sepsis Pipeline: Overview |
| 4. | Analytical Perspective In-depth Commercial Assessment |
| 5. | Sepsis Clinical Trial Therapeutics |
| 6. | Sepsis Pipeline: Late-Stage Products (Pre-registration) |
| 7. | Sepsis Pipeline: Late-Stage Products (Phase III) |
| 8. | Sepsis Pipeline: Mid-Stage Products (Phase II) |
| 9. | Sepsis Pipeline: Early-Stage Products (Phase I) |
| 10. | Sepsis Pipeline Therapeutics Assessment |
| 11. | Inactive Products in the Sepsis Pipeline |
| 12. | Company-University Collaborations (Licensing/Partnering) Analysis |
| 13. | Key Companies |
| 14. | Key Products in the Sepsis Pipeline |
| 15. | Unmet Needs |
| 16. | Market Drivers and Barriers |
| 17. | Future Perspectives and Conclusion |
| 18. | Analyst Views |
| 19. | Appendix |
For further information on the sepsis cure research, reach out @ Medication for Sepsis Treatment
Related Reports
Sepsis Market
Sepsis Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key Sepsis companies, including Regeneron Pharmaceuticals, Merck Sharp & Dohme LLC, Shionogi Inc., Sentien Biotechnologies, Inc., Basilea Pharmaceutica, Vivacelle Bio, SciClone Pharmaceuticals, Grifols, Asahi Kasei Pharma, Sanofi, BioAegis Therapeutics, Pharmazz, ARTCLINE GmbH, Revimmune SAS, Inotrem, Enlivex Therapeutics, Adrenomed, Tianjin Chasesun Pharmaceutical Co., LTD, Suzhou Yabao Pharmaceutical R&D Co., Ltd., Matisse Pharmaceuticals, CGE Healthcare, and others.
Sepsis Diagnostics Market
Sepsis Diagnostics Market Insights, Competitive Landscape, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key sepsis diagnostics companies, including BD, Cytovale™ Inc, Thermo Fisher Scientific Inc, Abbott, F. Hoffmann-La Roche Ltd, bioMérieux SA, Danaher, T2 Biosystems Inc, Luminex Corporation, Bruker, Immunexpress Inc, Axis-Shield Diagnostics Ltd, Quidel Corporation, Siemens Healthcare Private Limited, EKF Diagnostics Holdings plc, Seegene Inc, Boditech Med Inc, and others.
Uncomplicated Urinary Tract Infection Market
Uncomplicated Urinary Tract Infection Market Insight, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key uUTI companies, including GlaxoSmithKline, Iterum Therapeutics, Inmunotek, Janssen Pharmaceuticals, Fimbrion Therapeutics, and others.
Complicated Urinary Tract Infection Market
Complicated Urinary Tract Infection Market Insight, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key cUTI companies, including Spero Therapeutics, Wockhardt, Venatorx Pharmaceuticals, Allecra Therapeutics, Nabriva Therapeutics AG, MerLion Pharmaceuticals, and others.
Urinary Tract Obstruction Treatment Devices Market
Urinary Tract Obstruction Treatment Devices Market Insight, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key urinary tract obstruction treatment devices companies, including B. Braun Melsungen AG, Boston Scientific Corporation, Cook, Becton, Dickinson, and Company, Teleflex Incorporated, Olympus, Dornier Medtech, EMS Urology, Advin Health Care, Inceler Medikal Co. Ltd., STORZ MEDICAL AG, Shockwave Medical Inc., ACE Medical Devices Pvt. Ltd., Walz Elektronik GmbH, Applied Medical Resources Corporation, Richard Wolf GmbH, Stryker, Urocare Products, Inc., J and M Urinary Catheters LLC, Rocamed, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
CONTACT: Contact Us Shruti Thakur [email protected] +14699457679
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. NewIndiaObserver.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. NewIndiaObserver.com takes no editorial responsibility for the same.
